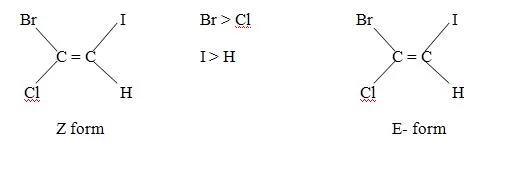
Cahn – Ingold – Prelog priority sequence can be usefully applied in giving names to such a structure. The group of highest priority, on both the double bonded carbon atom is first chosen. If the two group of highest priority are on the opposite side of the double bond. The alphabet E (from the German word Entagagen meaning opposite) is used for the structure and if the groups of highest priority are on the same side the alphabet Z (from German word, Zussamen meaning together) is used. Thus E stands for opposite side and Z for the same side. For example.
Generally cis isomer is said to be Z form and trans isomer is said to be E forms but due to sequence rule there are some exception. For example, 1,2 dichloro – 1 – bromoethene
Although Cl atom present on opposite side but according to sequence rule it is Z form.
Determination of the configuration of geometrical isomers
(i) Physical methods:- Generally trans-isomer has high melting point and low boiling point than cis isomer.
(ii) Solubility:- Solubility of cis isomer is more. For example, solubility of maleic acid is 3.0 g/100 mL of water at 293 K and solubility of fumaric acid is 0.7 g in 100 mL of water at 293 K.
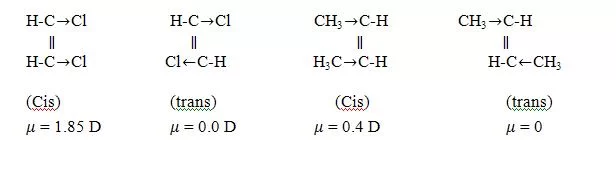
(iii) Dipole moment measurement:- The trans isomers are symmetrical and hence will have zero or low dipole moment as compared to the cis isomer.
μ = 0
(iv) Chemical method:- (method of cyclisation)
(i) In this method geometrical isomers may be obtained from cyclic compounds. If cyclic compound has two groups at cis and trans position than compound obtained will be in the similar position. For example, benzene or p-quinone on oxidation forms unsaturated dicarboxylic acid.
In benzene and p-quinone both hydrogen atoms are in cis position to double bond they will be at cis position in dicarboxylic acids and thus dicarboxylic acid is in cis form and another isomer will be trans (fumaric acid m.p. 575 K).
(ii) By changing geometrical isomer in cyclic compounds: A molecular containing two – COOH groups attached to different carbon atom will form an anhydride. If the groups are on same side i.e. the structure is cis then maleic acid forms anhydride easily and therefore it can be identified as cis compound. In fumaric acid the two – COOH groups are on the opposite side and therefore it is very difficult for them to interact under such conditions in which maleic acid does.
Frequently Asked Questions (FAQs)
Q.1. So why is the E/Z system preferred to cis/trans?
Ans. The cis/trans system can only do well when one has the same groups on both sides of the carbon. It is obscure in more complex molecules which have varied substituents. With priority rules, the E/Z system eliminates this ambiguity and is applicable on all alkenes.
Q.2. Priorities: How are substituents assigned?
Ans. The priority is based on the atomic number: the atom that has a higher atomic number is allocated a higher priority. In case of a tie, the successive atoms on the chain are compared until a difference is discovered.
Q.3. Is it possible to use E/Z nomenclature with other compounds than alkenes?
Ans. This can also be extended to other systems with limited rotation, like some cyclic compounds and molecules with double bonds with nitrogen (such as oximes).
Q.4. What would be the case when two groups attached on a carbon are the same?
Ans. When one of the two carbon atoms is substituted by two identical substituents, then that compound can not have geometrical isomers (and therefore E/Z nomenclature).
Q.5. Can a compound have more than one E/Z designation?
Ans. Yes, double and triple bonded compounds may contain more than one stereocenter which can each have its E or Z-configuration (e.g. E,Z or Z,Z).
Conclusion
The E/Z nomenclature system is an accurate and universally recognized system of nomenclature of geometrical isomers, where the older cis/trans system is unclear. According to the regulations of the Cahn-Ingold-Prelog priority rules, this system gives priorities to substituents attached to the double bonded carbon atoms. When the higher-priority groups are on the same side, the isomer is denoted by Z (the German word “zusammen”, meaning together), and when on different sides, denoted by E (the German word “entgegen”, meaning opposite).
This system is fundamental in organic chemistry as it brings simplicity and prevents confusion in case of complex molecules. The study of E/Z nomenclature assists chemists in describing molecular structures accurately, physical and chemical properties, and communicating effectively in the science domain.
Author
Dr. Ritu Jangid
Assistant Professor, Department Of Science
Biyani Group Of Colleges, Jaipur

