The splitting of molecular rotational energy levels in presence of external electric field (E) is known as the Stark effect after the physicist J. Stark who was awarded Nobel prize for his discovery of Doppler effect and splitting of spectral lines in electric field. Stark effect was first observed in atomic spectra. The rotational lines may be shifted as well as splitted (Fig. 14). Consider a rotating linear molecule with angular momentum perpendicular to the electric field, the field tends to twist the dipole and gives a faster rotation when the dipole is oriented in the direction of the field and slower rotation when it is opposite to the field. This minor difference between the dipoles pointing in two directions causes the splitting of energy levels.
It the dipole moment has a component and if the dipole momentum J, a first order Stark effect is observed and if the dipole moment is perpendicular to the angular momentum, a second order Stark effect is seen. A first order Stark effect applies when the splitting of rotational levels is directly proportional to the electric field. Symmetric top molecules, which have the component of dipole moment along the direction of the total rotational angular momentum, exhibit first order Stark effect. In second order Stark effect, the splitting of rotational levels by an electric field E is proportional to E2. This type of behavior is exhibited by Linear molecules where the dipole moments is perpendicular to the total angular momentum of the molecular rotation.
The shift of rotational frequency, ∆v, for a linear gaseous molecule is given by,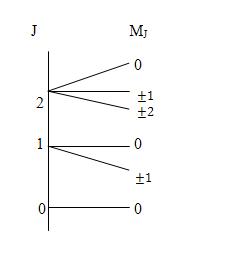
∆v ∝ Square of (u,E)
By knowing E and measuring ∆v, the electric dipole moments u can be calculated. Since the magnitude of splitting is dependent on the molecular dipole moment, so Stark effect permits a direct method for its accurate determination. The dipole moment of the molecule in a particular vibrational state could be measured rather than the average moment of all the states. The splitting under an effect field also depends on the rotational quantum number of the transition involved and it can be utilized to identify the rotational transitions. Stark effect splits the degeneracy of the J level into (2J + 1) levels and hence multiplet structure has been observed for all the lines with J > 0. Since the measurements can be made on gas samples at 10-3 torr, the measured dipole moment is accurate and not affected by molecular interactions and solvents effects.
Frequently Asked Questions (FAQs)
Q.1: What do you understand by the Stark effect?
Ans: It explains the coupling of external electric field and splitting or shifting spectral lines in
atoms / molecules.
Q.2: Who discovered the Stark effect?
A: The phenomenon is called the Stark effect after Johannes Stark who discovered it and won a
Nobel prize for his work.
Q.3: What is the difference between first-order and second-order Stark effects?
Ans: The splitting varies directly with field strength, a first-order effect. Instead, a second-order
effect scales with the strength of the field squared.
Q.4: What is the benefit of using the Stark effect to study molecules?
Ans: It enables accurate calculations of electric dipole moments and serves as a guide for
investigating certain rotational transitions in molecules.
Conclusion
The Stark effect provides a simple but profound method to investigate the response of individual molecules in an external electric field. The application of these fields results in splitting of the rotational energy levels, allowing scientists to gain valuable information about molecular structure and orientation of dipole moments. The first- or second-order nature of such splittings provides the ability to investigate rotational transitions extremely well.
The key advantage of this effect is its ability to both characterize the dipole moment of a molecule with great precision and consider not just an average over many vibrational states but at times within certain individual ones. As the tests are mostly performed within a low-pressure room, they produce neat and consistent data as there is less contamination from nearby molecules. Overall, the Stark effect remains a powerful technique in molecular spectroscopy which improves our understanding of how molecules behave and what properties they have.
Author:
Dr.Yogesh Kumar
Assistant Professor,Department of Science
Biyani Group of Colleges,Jaipur

